   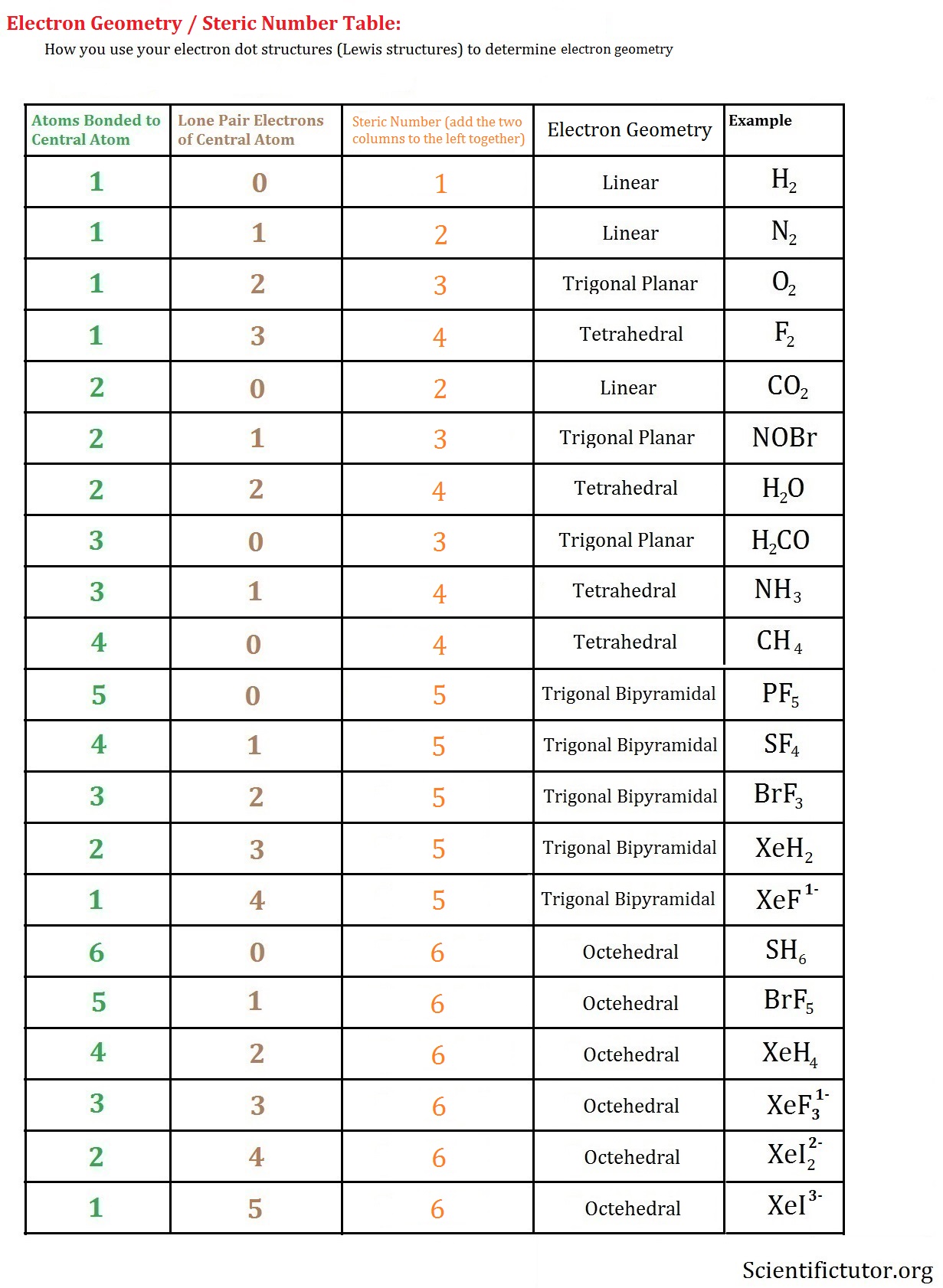
Therefore, there is a need to understand the molecular geometry of CH2F2. In brief, a carbon atom, a central atom, will form four single bonds without any lone pair on it.īut we cannot limit ourselves to the only Lewis structure for understanding the chemical reactions of CH2F2, which involves bond breaking and bond formation. Hence, there is a formation of the single bond between carbon and hydrogen as well as carbon and fluorine. In the Lewis structure of CH2F2, both Hydrogen and Fluorine atoms are sharing only one electron with the carbon atom. Therefore, the Lewis structure of CH2F2 can be represented as: The valence electron in carbon is four and hence, it will share its two valence electrons with two hydrogen atoms and the remaining two electrons with two fluorine atoms, resulting in the formation of an octet around it. Now, we have to arrange 20 valence electrons in the Lewis structure of CH2F2 with carbon as a central atom. The combining atom will achieve the nearest noble gas configuration by sharing electrons. The important conditions for the Lewis structures are that every combining atom has to contribute at least one electron for sharing with other atoms and a chemical bond is formed owing to the sharing of electron pairs between atoms. 
In the Lewis structure of the molecule, valence electrons are represented as dots. The total number of valence electrons in difluoromethane are 4 + 1(2) + 7(2) = 20 electrons. Therefore, carbon will be the central atom in difluoromethane. Hence, carbon can form four chemical bonds whereas hydrogen and fluorine can form only one chemical bond. The group valence indicates the number of chemical bonds an atom can form with other atoms. Group valence of carbon, hydrogen, and fluorine is 4, 1, and 1, respectively. Hence, the number of valence electrons in carbon, hydrogen, and fluorine is 4, 1, and 7, respectively. In CH2F2, carbon, hydrogen, and fluorine are from groups 14, 1, and 17, respectively. Only the valence electrons, which are present in the outermost shell of an atom participate in the bond formation. Kossel and Lewis were the first to explain the formation of chemical bonds in terms of electrons especially valence electrons.Īccording to Lewis, every atom tends to achieve a stable octet when they form chemical bonds except hydrogen and helium. The chemical bond is an attractive force between two atoms and atoms form a chemical bond to achieve stability as most of the elements are unstable in their atomic form. We will understand every feature in detail step by step. The key features of chemical bonding in difluoromethane are shown in the following table for quick learning. The low melting and boiling points of difluoromethane result in frostbite.ĬH2F2 contains fluorine, an electronegative element, but still, it does not show hydrogen bonding owing to the absence of an H-F bond. Its melting point and boiling points are -136 ☌ and -52 ☌, respectively. The molar mass of difluoromethane is 52.024 g/mol. It is utilized in the refrigeration, air conditioner, and fire extinguishers process due to its common use in endothermic processes. It has high thermal stability and slight solubility in water owing to its polar nature. It is a colorless gas at standard temperature and pressure. Haloalkanes or alkyl halides are organic compounds, which contain at least one halogen atom bonded to the carbon atom. CH2F2 or difluoromethane or difluoromethylene is an organic compound of the haloalkane family. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
